Message From The Chairman
 During the last decade, new and exciting changes have developed in the area of Physiology. In addition to a cadre of new investigative techniques, we have seen an explosion of interest in cellular and molecular research into physiological phenomena. Our Department has successfully blended our traditional systemic research approach with many new cellular and molecular approaches to research. In addition, we have developed strong research collaborations with clinical researchers to investigate physiological questions in areas such as hypertension, diabetes, cancer, heart disease, etc. Our Department has a strong interest in cardiovascular research and our efforts are potentiated by research interactions with both basic science departments and many clinical departments and subspecialties. The Department is located primarily in a modern 14 story research facility on the Health Sciences Campus. The Health Sciences Center is a well-integrated facility of both clinical and academic entities.
During the last decade, new and exciting changes have developed in the area of Physiology. In addition to a cadre of new investigative techniques, we have seen an explosion of interest in cellular and molecular research into physiological phenomena. Our Department has successfully blended our traditional systemic research approach with many new cellular and molecular approaches to research. In addition, we have developed strong research collaborations with clinical researchers to investigate physiological questions in areas such as hypertension, diabetes, cancer, heart disease, etc. Our Department has a strong interest in cardiovascular research and our efforts are potentiated by research interactions with both basic science departments and many clinical departments and subspecialties. The Department is located primarily in a modern 14 story research facility on the Health Sciences Campus. The Health Sciences Center is a well-integrated facility of both clinical and academic entities.
We are excited about the new and innovative research approaches being explored in our Department and in the field of Physiology. I, along with our faculty and graduate students would like to welcome you to our Website.
Irving G. Joshua, Ph.D.
Professor and Chairman
PHYSIOLOGY MARCH 2025 NEWSLETTER
MS Program
Items of Interest
Deadlines
MS application:
July 31, 2025 for Fall Semester 2025
*MS Program only begins in Fall
Please contact Mrs. Jennifer Wells at Jennifer.wells@louisville.edu when you have submitted your application to let the department know that you have applied.
PhD application:
The deadline for Ph.D. applications for Fall 2025 is January 31, 2025
2024-25 MS program will be in-person.
The PhD program will remain in-person.
Physiology Research Seminar Series Spring 2025
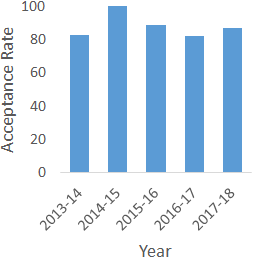
Professional school acceptance rate for students who have successfully completed the M.S. in Physiology at the University of Louisville
| Year | Acceptance Rate |
|---|---|
| 2013-14 | 83 |
| 2014-15 | 100 |
| 2015-16 | 89 |
| 2016-17 | 82 |
| 2017-18 | 87 |
 Facebook
Facebook Twitter
Twitter Linkedin
Linkedin