UofL equine business alumna Isabella Leslie: Bluegrass racetracks to Dubai and...
As anticipation builds for the 150th Kentucky Derby, many will be searching for the perfect hat to wear to Churchill Downs. Isabella Leslie, a...
UofL Rugby Club wins national championship
The University of Louisville Rugby Club has secured the Collegiate Rugby Championship Division 1 national title. They finished with a 4-0 record, outsourcing opponents 114-5,...
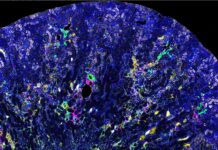
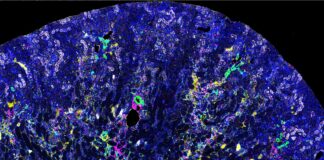
UofL researchers honored for groundbreaking study linking cancer and kidney disease
A team of researchers at the University of Louisville has been honored for their pioneering work in uncovering a new connection between cancer treatment...
Winners of 2024 Grawemeyer Award in Education discuss race and public university funding
University of California researchers Laura Hamilton and Kelly Nielsen, co-authors of the 2021 book, "Broke: The Racial Consequences of Underfunding Public Universities," presented key...
2024 Grawemeyer music award winner explains how music transcends language
For Aleksandra Vrebalov, visiting Louisville to give a public talk on “Missa Supratext,” her nontraditional choral work, was more than your typical lecture.
It was...
Psychologist Ann Masten talks about resilience during 2024 Grawemeyer Award lecture
Did you ever meet someone who not only survived, but thrived, despite a trauma-filled past or daunting obstacles? Exploring the human capacity to overcome...
2024 Grawemeyer Awardee in world order calls on military to reduce carbon emissions
What began as a simple search for data to support a presentation on climate change turned into an extensive project and a book calling...
2024 Grawemeyer prize winner in religion explores God’s humanity
The traditional story of the rainbow as a symbol of hope and God's unwavering love might be incomplete, according to Rev. Charles Halton, winner...